 the Henry's Law constant (in mol L kPa 1) for the solution of methane (CHag) and water (H2Ow) if 3.00g of methane gas is dissolved into 325mL of water, and the partial pressure of methane is measured as 0.5kPa. The value 55.85 g/mol means that a mole of iron atoms 6.02 x 10 23 (atoms) has a mass of 55.85 grams. In most periodic tables of the elements, the molar mass value is located in the top right hand corner of an elements cell. the partial pressure of CO) above a solution if 4.25g of CO) is dissoved in 100.0mL of solution at 25'C. This value has been calculated for each element and is usually included in a periodic table. Molar mass of K 39.1 g Molar mass of Mn 54.9 g Molar mass of O 16.0 g Molar mass of KMnO 4 39.1 g + 54.9 g + (16.0 g x 4) Molar mass of KMnO 4 158.0 g Use this number to convert grams to moles. 62.5mL of acetone (C3HsOu) dissolved into 125mL of 1-propanol (C H,Op) (acetone is a molecular liquid density of pure acetone 0.791g/mL density of pure 1-propanol 0.803g/mL) 2. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in HNO3: Molar Mass (g/mol) H (Hydrogen) 1 × 1.00794 1.00794. To convert grams to moles, the molar mass of the solute is needed, which can be found on certain periodic tables. (caffeine is a molecular solid, density of pure H-Op 1.00g/mL) d. 10.1mL of ethanol (C,HsOHp) dissolved into 230ml of H2Op density of pure HO-1.00g/ml) 100mg of caffeine (GHoN。2m) dissolved into 12.1L of H (ethanol is a molecular liquid density of pure ethanol 0.789g/mL c. Molarity is defined as the number of moles of solute in exactly 1 liter (1 L) of the solution: M molsolute Lsolution (1.4.3.4) (1.4.3.4) M m o l s o l u t e L s o l u t i o n. 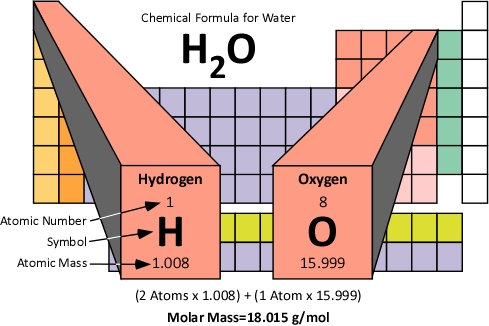
Lets practice determining the molar mass of some common substances. Molarity (M) is a useful concentration unit for many applications in chemistry. The periodic table lists the molar mass of each element. 
(K-COssy is an ionic solid density of pure Hy- 1.00g/ml) b. Avogadros number is 6.02 x 10 23 atoms per mole or molecules per mole. 
De termine the molarity, the molality, the mass percent, and the mole fraction of the solutes applicable): in the following mixtures (remember to account for ion dissociation when 3.3g of K Coss) dissolved into 500.mL of HzOp a. (If you are required to calculate the molar mass of a compound, use a periodic table that has ot least two decimal places for each element. The volume and molarity of the solution are specified, so the amount (mol) of solute is easily computed as demonstrated in Example 4.5.3: M molsolute Lsolution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
